TCVM Research Update: Evidence-Based Application of Acupuncture for Pain Management in Companion Animal Medicine
Posted by Dr. Luke on
Evidence-Based Application of Acupuncture for Pain Management in Companion Animal Medicine
1Essex Animal Hospital, 355 Talbot St N, Essex, ON N8M 2W3, Canada
2Chi University, 9650 W Hwy 318, Reddick, FL 32686, USA
3Arbor Pointe Veterinary Hospital, Animal Pain Center, 42043 Ford Rd, Canton, MI 48187, USA
*Author to whom correspondence should be addressed.
Vet. Sci. 2022, 9(6), 252; https://doi.org/10.3390/vetsci9060252
Received: 31 March 2022 / Revised: 3 May 2022 / Accepted: 18 May 2022 / Published: 26 May 2022
(This article belongs to the Special Issue Frontiers of Herbal Medicine and Acupuncture in Veterinary Medicine)
Abstract
The use of veterinary acupuncture for pain relief is expanding among small animal practitioners. Although acupuncture was developed as part of the medical system in Ancient China, research into the scientific basis of its effects is expanding rapidly. Acupuncture is very effective for analgesia on a local, segmental, and suprasegmental level. Many forms of acupuncture can be used independently or as part of a balanced multi-modal approach for the control of acute and chronic pain. In the hands of a skilled practitioner, acupuncture can be a safe and effective modality for treating pain in companion animals. This article outlines the mechanisms of action of acupuncture, its related neurophysiology and provides examples from the literature demonstrating its effectiveness.
1. Introduction
Traditional Chinese Medicine (TCM) has been practiced for more than 2000 years. It is based on the philosophy that wellness exists in balance and disease is caused by an imbalance of energies within the body. Acupuncture is one of branches of TCM and Traditional Chinese Veterinary Medicine (TCVM). It is a highly effective method to treat pain. [1,2]. The use of veterinary acupuncture for pain relief is expanding among small animal practitioners. It is used as an adjunct to conventional practices, as an alternative when conventional treatments fail to resolve the patient’s pain or when a client is seeking a drug-free way to mitigate pain [1,3,4,5]. The most recent canine and feline pain management guidelines, developed by the World Small Animal Veterinary Association Global Pain Council include acupuncture as a non-pharmaceutical treatment for small animal pain (https://wsava.org/global-guidelines/global-pain-council-guidelines, accessed on 21 November 2021).
Research into the scientific basis of acupuncture is expanding as evidenced by the growing number of scientific papers published on this topic. Of these roughly 4000 papers, 41% are focused on acupuncture for analgesia or pain relief [6] and the majority concerned humans or laboratory animals [7,8,9,10,11]. Nevertheless, these principles can be integrated into clinical veterinary practice. There have been some recent clinical studies in companion animals showing the effectiveness of acupuncture at mitigating pain [12,13,14,15,16,17,18].
Despite the increase in research and pet owner acceptance, some veterinary practitioners are reticent to integrate acupuncture into their practices because they believe it is based on an antiquated system [19]. TCVM acupuncture does use metaphorical language to describe patterns of disease, treatment, and pathophysiology; however, the terminology can be translated into physiological and biomedical principles that can be understood by the conventionally trained veterinarian. Acupuncture has evolved into a medically appropriate, scientifically driven treatment for pain in companion animals [3].
2. Acupuncture Points and Meridians
Acupuncture involves the stimulation of specific anatomical locations called acupuncture points or acupoints to create a local, segmental and general physiological effect that is mediated by neuromodulation [19,20]. Acupoints are neurovascular bundles consisting of a concentrated area of free nerve endings, lymphatics, small arterioles, venioles and mast cells. Areas that are richly innervated and areas with high numbers of somatic afferent fibers frequently contain many acupuncture points [19]. Acupuncture points are divided into Four Types (See Table 1) and have a higher electrical conductance, lower impedance and a higher capacitance than the surrounding tissue. Many of these points are located in palpable depressions and in areas where pain and muscle dysfunctions generate myofascial trigger points [21].
Table 1. Types of Acupuncture points (Data from Reference [21]).
Acupuncture points may be stimulated by the insertion of a tiny filiform needle, application of heat (moxibustion) or pressure (acupressure), use of laser light, friction and cupping. In addition, practitioners may elect to inject the acupoints with substances to induce a longer stimulation of the points. Examples of this are injections of vitamin B 12 or implantation of gold beads into acupuncture points [1,2]. Acupuncture needles may be stimulated manually by twisting or moving the needle during the treatment or may be stimulated by the use of electroacupuncture (EA). EA involves attaching an electrode to a pair of acupuncture points and running a small electric current through them. It is thought to provide a stronger and longer-lasting stimulation [1,2,21].
When a needle stimulates an acupuncture point, it activates neural and neuroactive components. These activated components that are found within the skin, connective tissue and muscle surrounding the inserted needed have been defined as neural acupuncture units (NAUs) [22]. With the variety of acupuncture points, it is important to balance any acupuncture treatment with local and remote points to address different sources of pain.
In TCVM, acupuncture points represent an area of concentrated Qi (energy or life force). The stimulation of these points encourages a smooth and harmonious flow of Qi along the meridians or channels, which link the acupuncture points throughout the body. There are 12 regular or principal meridians, which are named after Chinese or Zhang Fu organs. The Zhang Fu organs have Western names but are not considered true anatomical structures. Rather, they are a collection of organs that produce and regulate Qi. These organs have interconnected functions that explain how Qi is produced and flows through the body [23]. The blockage of Qi or nerve energy causes pain. The manipulation of one acupuncture point may cause an effect in an area remote to the site but further down the meridian. The Meridians have names such as Bladder and Gall Bladder and the acupuncture points assigned numbers along the meridian. In addition to the twelve regular Meridians, there are eight Extra Meridians, and these have specific functions. The eight Extra Meridians maintain the homeostatic balance in the body by storing, regulating, and receiving excess pathogenic Qi, balancing the metabolism and supporting blood, fluids and Qi [1,2].
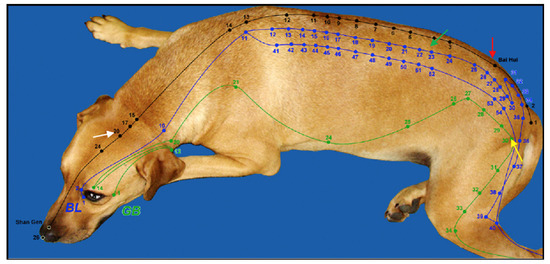
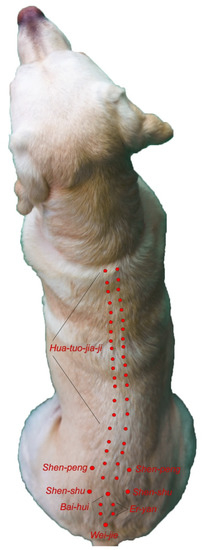
Extra meridian points are found on these Meridians and often have classical Chinese names such as Shen Shu. Both TCVM acupuncture and Western medical acupuncture use the same names for points to designate where the needles are placed [23]. See Figure 1 and Figure 2 for examples.
Figure 1. Meridians with Acupoints. Green is Gall Bladder (GB) Meridian, Blue is Bladder (BL) Meridian and Black is Governing Vessel (GV). GV-20 = White Arrow, Bl-23 = Green Arrow, Bai-Hui = Red Arrow, GB-30 = Yellow arrow. (Reprinted with permission from Ref [2]. 2003, H. Xie).
Whereas TCVM acupuncture looks at restoring the balance of Qi, Western Medical acupuncture looks at restoring physiologic homeostasis and utilizing mechanisms within an animal to relieve pain [24]. From this perspective, meridians are thought to be consistent with the peripheral nervous system, run along fascial planes, involve the lymphatics and may involve the interstitial space [25]. Physiologically, all acupuncture points share the same effects including fibroblast mechanotransduction, fascial interaction, neuromodulation leading to analgesia, fluid motion modulation and the manipulation of the microvascular environment. [23,26,27,28]. Some points may specifically cause immune modulation, affect the autonomic nervous system, visceral organs, or the brain, modulate lymphatic flow or cause neuromuscular effects [19].
Figure 2. Extra Meridian Points with Classical Chinese Names. Reprinted with permission from Ref. [29]. 2018, Dewey C and Xie S.
3. Analgesic Effects of Acupuncture
3.1. The Pain Pathway
To better understand how acupuncture is effective in treating pain, it is helpful to review the physiology of pain. The pain pathway, which includes transduction, transmission, modulation and perception is part of the physiology of pain. The first step in the pain pathway occurs when damaged cells release inflammatory mediators such as interleukins (IL), prostaglandins (PG), arachidonic acid (AA), serotonin, substance P, bradykinin, and glutamate. Transduction occurs when nociceptors convert this chemical signal into an electrical impulse [30].
Transmission involves sending an electrical impulse to the brain via the afferent nerve fibers. The fibers involved are the A-beta, A-delta and C fibers. See Table 2 for a comparison of the different types of nerve fibers. A-beta fibers are responsible for low-threshold or non-painful information, whereas A-delta fibers are responsible for the withdrawal reflex and sharp acute pain. C fibers, which transmit their information at a slower rate, are responsible for slow, burning pain which can intensify. The stimulation of C fibers has been implicated in the development of chronic pain [30].
Table 2. Characteristics of Nerve Fibers (Data adapted from Ref. [30]).
Modulations in the nerve signal occur in the dorsal horn (DH) of the spinal cord. The impulses from the A-Delta and C fibers change in the DH and are either inhibited or amplified. Excitatory and inhibitory neurotransmitters transform the signal before it reaches the brain. Transmitters such as glutamate intensify the pain signal whereas gamma-amino butyric acid (GABA) acts as an inhibitor. Serotonin can suppress the action of glutamate while enhancing that of GABA. Endogenous opioids are also released and influence the pain signal. Opioid receptors are triggered to suppress excitatory neurotransmission. A-beta nerve fibers also act as a gate control to suppress pain signals in the DH [30].
Perception is the final step in the pain pathway, and it involves the recognition and integration of pain. Multiple brain regions and ascending pathways are involved in the processing of pain. The perception centers in the brain control the overall autonomic, emotional and physical response to pain [30].
Acupuncture analgesia can be divided into local, segmental (spinal) or suprasegmental effects [21]. Local effects are mediated through the sensory nociceptors stimulating the afferent nerves whose cell bodies are in the dorsal root ganglia. Spinal effects are mediated by neurons in the dorsal gray matter of the spinal cord. These are second-order neurons which provide the ascending nociceptive tracts to the brain. In the brain, a descending pain-mitigating system (suprasegmental) can be stimulated to suppress the pain signal sent by the second-order neurons, thus providing analgesia [21].
3.2. Local Effects of Acupuncture
Local analgesic effects start immediately at the site of needle insertion and act as a form of counter-irritation. Collagen and elastin fibers wind around the needle and this acts on fibroblasts as well as local nerve endings and vasculature. The physical sensation of the needle causes the fibroblasts to release biochemical substances that elicit a response (mechanotransduction) [27,28]. Data suggest that the deeper the needle is inserted, the more dramatic the effects [31]. Neuronal signalling and needle insertion are intimately connected. When the fascia and tissue matrix is deformed by the needle, the local axon reflex is triggered in the nerve endings. This response is thought to cause the De Qi sensation which humans describe as dull or achy and travels from the site of initial needle insertion to the nerves. The De Qi response is associated with the effectiveness of the treatment and leads to the release of neuropeptides such as calcitonin gene-related peptide, nerve growth factor, substance P and many others [19,32].
The needle creates microtrauma or local tissue damage which activates Hageman’s tissue factor XII. This in turn results in the activation of the local coagulation cascade and the complement cascade, leading to the production of plasminogen, protein kinins, and prostaglandins. The microtrauma also causes mast cell degranulation, which releases histamine, heparin, proteases, and bradykinin. These local reactions ultimately result in increased blood flow to the area and local immune responsiveness, which help to relieve pain and reduce inflammation and edema. Connective tissues become stretched when needles are placed in acupoints, which in turn relaxes the muscles and tissues in the local area [19,32].
Many of the local effects of acupuncture are thought to be mediated by endogenous opioids. Tissue lymphocytes, macrophages and granulocytes release endogenous opioids when stimulated by an acupuncture needle. The peripheral sympathetic nerve receptors are acted on by these opioids to suppress the tissue nociceptors and to release more endogenous opioids into the area [19]. Studies in both humans and laboratory animals have shown increased levels of endogenous opioids in the plasma and CSF after acupuncture treatments [32]. A pilot study in dogs undergoing ovariohysterectomies compared dogs that received electroacupuncture for pain control versus those who received butorphanol. Both treatments were effective for pain control and the acupuncture group did not require any rescue pain control, although the butorphanol group did [33].
Other effects observed as a result of acupoint stimulation include an increase in cannabinoid CB2 receptors, which causes the upregulation of endogenous opioids, inhibition of cyclooxygenase-2 and prostaglandin production, and upregulation of TRPV-1 receptors in peripheral nerve endings [19]. A cascade of local and systemic effects ultimately provides analgesia to the patient for painful conditions. Painful local clinical conditions such as burns, lick granulomas, chronic wounds and local soft tissue injuries can benefit from the local analgesic effects of acupuncture [19].
3.3. Spinal or Segmental Effects of Acupuncture
Acupuncture-based modulation of pain is complex and involves many ascending and descending pain pathways, receptors, and molecules [19,32]. The gate control theory proposed by Melzack and Wall in 1965 is the main theory relating to the segmental mechanism of pain relief and explains pain modulation in the spinal cord [34]. The pain gate is located in the dorsal horn of the spinal cord. The substansia gelatinosa (SG) cells found in the dorsal horn control the transmission of pain impulses from the periphery to the CNS. Non-noxious stimulus can occur by needling or manual pressure, which stimulates the A-beta fibers and effectively closes the SG pain gates to painful input and prevent the pain signal from travelling further up to the CNS [34]. The main structures in the spinal cord that are involved in segmental pain relief are the A-Delta and C-ascending pain fibers and the A-beta fibers which effectively close the pain gate. See Table 1. Gate control theory says that a high volume of input from the A-Beta fibers effectively closes the gate in the SG to the pain impulses carried by the C and A-Delta fibers [19].
In addition to the gating of the nerve fibers in the spinal cord, other mechanisms and molecules are involved in segmental pain relief. N-methyl-d aspartate (NMDA) is a receptor for the excitatory neurotransmitter glutamate which is released with the stimulation of nociceptors. The activation of the NMDA receptors in the DH has been associated with hyperalgesia and neuropathic pain. Acupuncture has been shown to decrease the NMDA receptor activity in the dorsal horn neurons by increasing the levels of serotonin, noradrenaline, and endogenous opioids [19,32,35,36,37]. Increased serotonin levels in the spinal cord enhance the ability of gamma aminobutyric acid (GABA) to suppress the transmission of the pain signal [19,35]. Acupuncture stimulation reduces spinal glial cell activation and suppresses the release of inflammatory cytokines such as Il-1 B, IL-6, TNF a, COX-2 and PGE2 [19]. Substance P, which activates glial cells, is also suppressed by acupuncture while GABA release is facilitated by acupuncture. Increased GABA in the extracellular space is associated with anti-nociceptive activity. Just as in the periphery, acupuncture decreases the expression of COX-2 in the spinal cord, thereby reducing wind up pain [19,35].
Spinal segmental dysfunction is a term used by medical acupuncturists, physiatrists, physiotherapists, chiropractors and osteopaths. It has been defined as a functional problem in the somatic system, which may include skeletal, arthrodial, or myofascial structures and their related vessels, lymphatics or nerves [38]. The spinal cord segments and vertebral column are closely related with the roots and spinal nerves distributed throughout the vertebral column. Dermatomes exist for each spinal nerve and studies have shown that there is a considerable overlap of up to three dorsal roots in the lumbosacral area as well as an overlap of the cutaneous area of peripheral nerves. When treating these segments with acupuncture, any part of one segment will treat the entire segment [32]. Segmental dysfunction is a significant cause of pain in small animals and affects all components of the musculoskeletal system. It is not a true disease but often manifests as weakness, asymmetry, tenderness, decreased range of motion in joints, myofascial pain, and pathologic muscle tone. There is some evidence that segmental dysfunction is mediated by the sympathetic nervous system [32].
The segmental effects of acupuncture extend to visceral analgesia as well as musculoskeletal and neurological analgesia. Visceral and somatic afferent fibers converge in the dorsal horn. According to de Lahunta, visceral referred pain is “referred to the surface of the body innervated by the general somatic afferent (GSA) neurons whose axons terminate in the same spinal cord segment and on the same neuronal cell bodies as the general visceral afferent neurons” [39]. If the appropriate somatic receptors at the segmental level are stimulated, then visceral pain can be suppressed.
Acupuncture has been shown to alter blood flow and autonomic activity that regulates visceral pain and function. Functional gastrointestinal disorders such as gastrointestinal motility and stomach acid secretion can be altered by acupuncture via somatosympathetic or somatoparasympathetic pathways depending on the acupoints stimulated [32].
3.4. Suprasegmental (Brain) Effects of Acupuncture
The effects of acupuncture on pain perception in the brain of companion animals are not as well understood as the segmental effects. Endogenous pain management primarily occurs through the stimulation of an anti-nociceptive network that projects to the dorsal horn from several different brain segments [19,31]. According to Dewey and others, the brain segments involved include the cerebral cortex, the hypothalamus, the periaqueductal gray (PAG) matter, the pons and parts of the medulla [19,32].
Chemical messengers involved in the suppression of pain perception include Beta endorphins, enkephalin, noradrenaline, dopamine and serotonin. Part of this network also includes the pituitary gland, which releases oxytocin and adrenocorticotrophic hormones, both of which have analgesic effects [20]. Human magnetic resonance imaging (MRI) studies have shown that when specific acupuncture points are stimulated, specific regions of the brain are activated or deactivated. Functional magnetic resonance imaging (fMRI) measures brain activity by detecting changes in blood oxygenation and flow that occur in response to neural activity. Several fMRI studies have shown that the limbic system, hypothalamus, and arcuate nucleus are all stimulated by acupuncture [40,41,42]. When the hypothalamus is stimulated by acupuncture, it releases Beta endorphins and in turn activates the PAG which has been shown to be the primary CNS structure responsible for descending pain inhibition [19,32]. Needling limb points has been shown to activate the intended brainstem center without producing a segmental effect, whereas needling a dermatomal segment such as a back Shu point for thoracolumbar pain produces a mainly segmental response or a segmental and suprasegmental response (descending brainstem response) [19]. A combination of different points can recruit local, segmental, and suprasegmental responses from the nervous system for optimum results for pain control.
Chronic stimulation can cause the CNS to alter its chemical and structural organization. This is called neuroplasticity. Maladaptive neuroplasticity can occur as a result of chronic pain that induces the CNS to facilitate pain transmission. However, acupuncture induces beneficial neuroplasticity that prevents or reverses the changes caused by chronic neuropathic pain. This neuroprotective effect is achieved via neurotransmitters, and by decreasing microglial activation and inflammation [19]. Analgesic effects produced by EA tend to be more potent and long lasting than those produced by DN alone. Low frequency EA (2–10 Hz) causes the release of endorphins and enkephalins, which are most effective for mitigating pain. Other pain relievers such as dynorphin and serotonin are released at higher frequencies and may impart an additive effect [16,19,43,44].
4. Clinical Applications of Acupuncture for Pain Mitigation
Acupuncture can be very effective in treating the chronic pain and decreased mobility of degenerative joint disease. The segmental effects of acupuncture can provide pain relief, improve circulation and healing, and increase mobilization [19]. Pain from stifle disease is modulated through the nerves at the L4-L6 spinal segments. When this area, as well as the muscles around the stifle are treated with dry needle acupuncture (DN), the stifle experiences significant pain relief [32]. This effect can be enhanced by the addition of electroacupuncture (EA). A retrospective study of senior dogs with cranial cruciate ligament (CCL) ruptures showed that those treated with acupuncture and Chinese Medicine recovered their stifle function within 6 to 10 months of initiating treatment, even though no surgery was performed [45]. In another study of 40 dogs with CCL disease, half of the dogs were assigned to receive acupuncture and TCVM therapy whereas the other half received conservative therapy with pain management, joint supplements and exercise therapy. Both groups of dogs showed improvements by week 24 but those treated with acupuncture attained quicker results [46].
Hip osteoarthritis (OA) is a common painful and chronic disease in medium to large breed dogs that can result from age related changes, or secondary to a congenital condition such as hip dysplasia. It is associated with significant pain and mobility issues and affects the quality of life of patients who suffer from this condition. Many clinical trials have indicated acupuncture to be useful for analgesia for these patients [47,48,49,50]. In addition to DN and EA, aquapuncture (acupoint injection) or the implantation of gold beads or wires was commonly performed [37]. In a Brazilian clinical study, nine dogs with confirmed hip dysplasia that had a poor response to medical therapy were injected either with the autologous stromal vascular fraction (SVF, n = 4) or allogeneic cultured adipose-derived stem cells (ASCs, n = 5) into three acupuncture points (BL-54, GB-29 and GB-30) near the affected joint. After 1 week, both groups demonstrated a marked improvement in lameness and pain scores which continued to improve up to day 30. Only one dog in the ASC group did not improve. This study showed that the acupoint injection of stems cells can be an effective therapy for the pain of hip OA [47].
Several studies and clinical case reports were conducted on gold bead implantation into acupuncture points around the hip for the treatment of canine hip dysplasia [48,49,50,51]. Some of these studies [48,49,50] showed positive results, although a German review showed that some of the research may have been flawed [51]. Objective outcome measures were used in one study; however, this study did not show any significant difference between the placebo group and the group treated with gold bead implantation [37].
Another interesting study compared EA and laser acupuncture (LA) for the treatment of coxofemoral OA. A total of 31 dogs were randomly assigned to two groups. The Canine Brief Pain Inventory (CBPI) was completed by the owner and the goniometric range of motion (ROM) of both coxofemoral joints was determined as the baseline for all dogs. One group received laser acupuncture with a class IV laser to hip points (BL-54, GB-29 and GB-30). A total of 12 joules (J) was delivered at each AP point. The other group received electroacupuncture to the same group of points. Each pet was treated once a week and only the owner was blinded to which therapy their pet was receiving. At the end of 8 weeks, the CBPI score was repeated by the owner while the researcher repeated the ROM measurements. Both groups showed an improvement in pain scores with the EA group having improved statistically more, whereas only the LA group showed an improvement in hip extension. Laser acupuncture appears to be a valid pain therapy for those dogs who are needle phobic [16].
Acupuncture, particularly EA, has been shown to improve analgesia and accelerate the recovery of motor functions in dogs with intervertebral disc disease (IVDD) [43]. Hayashi and colleagues conducted a study on 50 dogs with thoracolumbar IVDD of various grades. The dogs were randomised to receive either conventional medical treatment or conventional medical treatment with EA. The overall success rate, as determined by their ability to walk without assistance, was significantly higher with EA and the time to ambulation was also significantly shorter in the EA group [44]. Another study of dogs with thoracolumbar disc disease showed that when electroacupuncture was combined with Western treatment, the medium time to ambulation was shorter in those dogs that received acupuncture than in those dogs who did not [52]. A similar result was reported by Han and colleagues for a group of 80 paraplegic dogs in which half of the dogs were treated with prednisone and the remainder were treated with prednisone combined with acupuncture and electroacupuncture. The dogs who were treated with a combination of DN, EA and prednisone had significantly less pain, recovered to ambulation at a faster rate and had decreased relapses [53]. Supplementary Table S1 lists common acupuncture pain points that may be of interest to the reader.
5. Acupuncture Contraindications and Precautions
Acupuncture is a very safe modality. Adverse events can generally be avoided through careful patient selection. Placing needles in inflamed or infected skin should be avoided. Needles should not be used in patients with bleeding disorders. In pregnant animals, points that may cause premature labor should be avoided. EA should not be applied across tumor sites, across the chest in animals with pacemakers or across the skull in animals with seizure conditions. In areas of fracture or acute trauma, needle placement should be avoided. Although a broken acupuncture needle is rare, it is possible that a migrating, broken needle could cause harm [2].
6. Conclusions
Acupuncture has profound effects on both the peripheral and central nervous systems. There is plenty of research concerning laboratory animals and humans, but the field of veterinary acupuncture would benefit from more validated clinical studies in companion animals. For many veterinary patients, pain management can be challenging due to medical co-morbidities and the sensitivity of many patients to pharmaceutical pain control. Acupuncture fits well into a multi-modal model for the management of acute and chronic pain and is a safe complementary modality that can be embraced by trained practitioners in companion animal practices.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/vetsci9060252/s1, Table S1: Commonly used Pain Points. Reference [4] is cited in the supplementary materials.
Author Contributions
Conceptualization, J.L.H.; Writing—original draft preparation, J.L.H.; Writing—review and editing, J.L.H. and M.C.P. All authors have read and agreed to the published version of the manuscript.
Funding
This review paper received no funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Xie, H.; Wedemeyer, L. The Validity of Acupuncture in Veterinary Medicine. Am. J. Tradit. Chin. Vet. Med. 2012, 7, 35–43. [Google Scholar]
- Xie, H.; Preast, V. Xie’s Veterinary Acupuncture; Blackwell Publishing: Ames, IA, USA, 2007. [Google Scholar]
- Epstein, M.E.; Rodanm, I.; Griffenhagen, G.; Kadrlik, J.; Petty, M.; Robertson, S.; Simpson, W. 2015 AAHA/AAFP Pain Management Guidelines for Cats and Dogs. J. Am. Anim. Hosp. Assoc. 2015, 17, 251–272. [Google Scholar] [CrossRef]
- Cantwell, S.L. Traditional Chinese Veterinary Medicine: The Mechanism and Management of Acupuncture for Chronic Pain. Top. Companion Anim. Med. 2010, 25, 53–58. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shmalberg, J.; Memon, M.A. A Retrospective Analysis of 5195 Patient Treatment Sessions in an Integrative Veterinary Medicine Service: Patient Characteristics, Presenting Complaints, and Therapeutic Interventions. Vet. Med. Int. 2015, 2015, 983621. [Google Scholar] [CrossRef][Green Version]
- Han, J.S. Acupuncture analgesia: Areas of consensus and controversy. Pain 2011, 152 (Suppl. 2), S41–S48. [Google Scholar] [CrossRef]
- Berman, B.M.; Langevin, H.M.; Witt, C.M.; Dubner, R. Clinical Therapeutics Acupuncture for Chronic Low Back Pain. N. Eng. J. Med. 2010, 363, 454–461. [Google Scholar] [CrossRef][Green Version]
- Kaptchuk, T.J.; Chen, K.J.; Song, J. Recent clinical trials of acupuncture in the West: Responses from the practitioners. Chin. J. Integr. Med. 2010, 16, 197–203. [Google Scholar] [CrossRef][Green Version]
- Selmer, M.; Shiau, D.S. Therapeutic Results of Integrative Medicine Treatments Combining Traditional Chinese with Western Medicine: A Systematic Review and Meta-Analysis. Am. J. Tradit. Chin. Vet. Med. 2019, 14, 41–47. [Google Scholar]
- Chen, S.; Wang, S.; Rong, P.; Wang, J.; Qiao, L.; Feng, X.; Liu, J.; Zhang, J. Acupuncture for visceral pain: Neural substrates and potential mechanisms. Evid.-Based Complementary Altern. Med. 2014, 2014, 609594. [Google Scholar] [CrossRef][Green Version]
- Fan, A.Y.; Ouyang, H.; Qian, X.; Wei, H.; Wang, D.D.; He, D.; Tian, H.; Gong, C.; Matecki, A.; Alemi, S.F. Discussions on real-world acupuncture treatments for chronic low-back pain in older adults. J. Integr. Med. 2019, 17, 71–76. [Google Scholar] [CrossRef]
- Lane, D.M.; Hill, S.A. Effectiveness of combined acupuncture and manual therapy relative to no treatment for canine musculoskeletal pain. Can. Vet. J. 2016, 57, 407–414. [Google Scholar] [PubMed]
- Liu, C.M.; Chang, F.C.; Lin, C.T. Retrospective study of the clinical effects of acupuncture on cervical neurological diseases in dogs. J. Vet. Sci. 2016, 17, 337–345. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Silva, N.E.O.F.; Luna, S.P.L.; Joaquim, J.G.F.; Coutinho, H.D.; Possebon, F.S. Effect of acupuncture on pain and quality of life in canine neurological and musculoskeletal diseases. Can. Vet. J. 2017, 58, 941–951. [Google Scholar]
- Janssens, L.A.A. Observations on acupuncture therapy of chronic osteoarthritis in dogs: A review of sixty-one cases. J. Small Anim. Pract. 1986, 27, 825–837. [Google Scholar] [CrossRef]
- Chomsiriwat, P.; Ma, A. Comparison of the effects of electro-acupuncture and laser acupuncture on pain relief and joint range of motion in dogs with coxofemoral degenerative joint disease. Am. J. Tradit. Chin. Vet. Med. 2019, 14, 11–20. [Google Scholar]
- Shmalberg, J.; Burgess, J.; Davies, W. A randomized controlled blinded clinical trial of electro-acupuncture administered one month after cranial cruciate ligament repair in dogs. Am. J. Tradit. Chin. Vet. Med. 2014, 9, 41–43. [Google Scholar]
- Sousa, N.R.; Luna, S.P.; Cápua, M.L.; Lima, A.F.; Oliveira, F.A.; Viveiros, B.M.; Barbosa, L. Analgesia of preemptive pharmacopunc ture with meloxicam or aquapuncture in cats undergoing ovariohysterectomy. Ciência Rural. 2012, 42, 1231–1236. [Google Scholar] [CrossRef][Green Version]
- Dewey, C.W.; Xie, H. The scientific basis of acupuncture for veterinary pain management: A review based on relevant literature from the last two decades. Open Vet. J. 2021, 11, 203–209. [Google Scholar] [CrossRef]
- Gong, C.; Liu, W. Advanced research validates classical principles-the neurobiological mechanisms involved in acupuncture for pain management. Longhua Chin. Med. 2019, 2, 16. [Google Scholar] [CrossRef]
- Schoen, A.M.; Janssens, L.; Rogers, P.A.M. Veterinary Acupuncture. In Seminars in Veterinary Medicine and Surgery (Small Animal); American Veterinary Publicationsl: New York, NY, USA, 1986; Volume 1, pp. 224–229. [Google Scholar]
- Zhang, Z.J.; Wang, X.M.; McAlonan, G.M. Neural acupuncture unit: A new concept for interpreting effects and mechanisms of acupuncture. Evid.-Based Complementary Altern. Med. 2012, 2012, 429412. [Google Scholar]
- Wright, B.D. Acupuncture for the treatment of animal pain. Vet. Clin. Small Anim. Pract. 2019, 49, 1029–1039. [Google Scholar] [CrossRef]
- Lu, D.P.; Lu, G.P. An Historical Review of Perspective on the impact of acupuncture on US Medicine and Society. Med. Acupunct. 2013, 25, 311–316. [Google Scholar] [CrossRef][Green Version]
- Zhang, W.B.; Jia, D.X.; Li, H.Y.; Wei, Y.L.; Yan, H.; Zhao, P.N.; Gu, F.F.; Wang, G.J.; Wang, Y.P. Understanding qi running in the meridians as interstitial fluid flowing via interstitial space of low hydraulic resistance. Chin. J. Integr. Med. 2018, 24, 304–307. [Google Scholar] [CrossRef] [PubMed]
- Wright, B.D. Acupuncture for the Treatment of Neuromuscular Conditions in Dogs and Cats, with Emphasis on Intervertebral Disc Disease. J. Am. Holist. Vet. Med. Assoc. 2021, 63, 23–33. [Google Scholar]
- Langevin, H.M. Acupuncture, connective tissue, and peripheral sensory modulation. Crit. Rev. Eukaryot. Gene Expr. 2014, 24, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Nedergaard, M.; Howe, A.K. Cellular control of connective tissue matrix tension. J. Cell. Biochem. 2013, 114, 1714–1719. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dewey, C.W.; Xie, H. Clinicians Guide to Canine Acupuncture; Chi Institute Press: Reddick, FL, USA, 2018. [Google Scholar]
- Weish, A.J.; Yaksh, T. Nociception and Pain Mechanisms. In The Handbook of Veterinary Pain Management; Gaynor, J., Muir, W.W., Eds.; Elsevier Mosley: Saint Louis, MO, USA, 2015; pp. 10–41. [Google Scholar]
- Zhang, J.H.; Cao, X.D.; Lie, J.; Tang, W.J.; Liu, H.Q.; Fenga, X.Y. Neuronal specificity of needling acupoints at same meridian: A control functional magnetic resonance imaging study with electroacupuncture. Acupunct. Electro-Ther. Res. 2007, 32, 179–193. [Google Scholar] [CrossRef]
- Fry, L.M.; Neary, S.M.; Sharrock, J.; Rychel, J.K. Acupuncture for analgesia in veterinary medicine. Top. Companion Anim. Med. 2014, 29, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Groppetti, D.; Pecile, A.M.; Sacerdote, P. Effectiveness of electroacupuncture analgesia compared with opioid administration in a dog model: A pilot study. Br. J. Anaesth. 2011, 107, 612–618. [Google Scholar] [CrossRef][Green Version]
- Melzack, R.; Wall, P.D. Pain mechanisms: A new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lao, L.; Ren, K.; Berman, B.M. Mechanisms of acupuncture-electroacupuncture on persistent pain. Anesthesiology 2014, 120, 482–503. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhao, Z.Q. Neural mechanism underlying acupuncture analgesia. Prog. Neurobiol. 2008, 85, 355–375. [Google Scholar] [CrossRef]
- Xie, H.; Sivula, N. Review of Veterinary Acupuncture Clinical Trials. Am. J. Tradit. Chin. Vet. Med. 2016, 11, 49–60. [Google Scholar]
- Watkin, H. Segmental dysfunction. Acupunct. Med. 1999, 17, 118–122. [Google Scholar] [CrossRef]
- de Lahunta, A.; Glass, E. Veterinary Neuroanatomy and Clinical Neurology, 3rd ed.; Saunders Elsevier: St. Louis, MO, USA, 2009; p. 540. [Google Scholar]
- Kong, J.; Kaptchuk, T.J.; Polich, G.; Kirsch, I.; Vangel, M.; Zyloney, C.; Rosen, B.; Gollub, R.L. An fMRI study on the interaction and dissociation between expectation of pain relief and acupuncture treatment. Neuroimage 2009, 47, 1066–1076. [Google Scholar] [CrossRef][Green Version]
- Kong, J.; Kaptchuk, T.J.; Polich, G.; Kirsch, I.; Vangel, M.; Zyloney, C.; Rosen, B.; Gollub, R.L. Expectancy and treatment interactions: A dissociation between acupuncture analgesia and expectancy evoked placebo analgesia. Neuroimage 2009, 45, 940–949. [Google Scholar] [CrossRef][Green Version]
- Kong, J.; Gollub, R.L.; Rosman, I.S.; Webb, J.M.; Vangel, M.G.; Kirsch, I.; Kaptchuk, T.J. Brain activity associated with expectancy-enhanced placebo analgesia as measured by functional magnetic resonance imaging. J. Neurosci. 2006, 26, 381–388. [Google Scholar] [CrossRef][Green Version]
- Roynard, P.; Lauren, F.; Xie, H.; Fowler, M. Acupuncture for Small Animal Neurologic Disorders. Vet. Clin. Small Anim. Pract. 2018, 48, 201–219. [Google Scholar] [CrossRef]
- Hayashi, A.M.; Matera, J.M.; de Campos Fonseca Pinto, A.C. Evaluation of electroacupuncture treatment for thoracolumbar intervertebral disk disease in dogs. JAVMA 2007, 231, 913–918. [Google Scholar] [CrossRef]
- Lee, L. Non-Surgical Treatment for Cranial Cruciate Ligament Rupture in Senior Dogs: A Retrospective Case Series. Am. J. Tradit. Chin. Vet. Med. 2019, 14, 49–64. [Google Scholar]
- Ciolanescu, B. Comparison of the Efficacy of Traditional Chinese Veterinary Medicine Versus Conservative Management for Treatment of Cranial Cruciate Ligament Injury in 40 Companion Dogs. Am. J. Tradit. Chin. Vet. Med. 2020, 15, 23–34. [Google Scholar]
- Marx, C.; Silveira, M.D.; Selbach, I.; da Silva, A.S.; Braga, L.M.; Camassola, M.; Nardi, N.B. Acupoint injection of autologous stromal vascular fraction and allogeneic adipose-derived stem cells to treat hip dysplasia in dogs. Stem Cells Int. 2014, 2014, 391274. [Google Scholar] [CrossRef][Green Version]
- Jaeger, G.T.; Larsen, S.; Søli, N.; Moe, L. Double blind, placebo-controlled trial of the pain-relieving effects of the implantation of gold beads into dogs with hip dysplasia. Vet. Rec. 2006, 158, 722–726. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, G.T.; Larsen, S.; Søli, N.; Moe, L. Two years follow-up study of the pain-relieving effect of gold bead implantation in dogs with hip-joint arthritis. Acta Vet. Scand. 2007, 40, 9. [Google Scholar] [CrossRef][Green Version]
- Bartholome, M.; Schulze, E. Efficacy of Gold Bead Acupuncture Treatment of Chronic Hip, Elbow and Spine Pain in 79 Dogs Following Unsuccessful Conventional Treatments. Am. J. Tradit. Chin. Vet. Med. 2020, 15, 17–30. [Google Scholar]
- Deisenroth, A.; Nolte, I.; Wefstaedt, P. Use of gold Implants as a treatment of pain related to canine hipdysplasia—a review. Part 2: Clinical trials and case reports. Tierarztl. Prax. Ausg. K Kleintiere/Heimtiere 2013, 41, 244–254. [Google Scholar] [CrossRef]
- Joaquim, J.G.; Luna, S.P.; Brondani, J.T.; Torelli, S.R.; Rahal, S.C.; de Paula Freitas, F. Comparison of decompressive surgery, electroacupuncture, and decompressive surgery followed by electroacupuncture for the treatment of dogs with intervertebral disk disease with long standing severe neurologic deficits. J. Am. Vet. Med. Assoc. 2011, 236, 1225–1229. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Han, H.J.; Yoon, H.Y.; Kim, J.Y. Clinical Effect of additional electroacupuncture on thoracolumbar intervertebral disc herniation in 80 paraplegic dogs. Am. J. Chin. Med. 2010, 38, 1015–1025. [Google Scholar] [CrossRef]
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
|
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
MEDICAL DISCLAIMER
This article is cited from Vet. Sci. 2022, 9(6), 252; https://doi.org/10.3390/vetsci9060252
The information mentioned in this article is for education and information only. It is not intended to diagnose, treat, cure, or prevent any disease and do not replace medical advice. Advice on treatment or care of an individual patient should be obtained through consultation with a physician or trained health care practitioner who has examined that patient or is familiar with that patient's medical history.
This information and advice published or made available through this website are not intended to replace the services of a physician or a health care professional acting under a physician’s supervision, nor does it constitute a doctor-patient relationship. Each individual’s treatment and/or results may vary based upon the circumstances, the patients’ specific situation, as well as the health care provider’s medical judgment and only after further discussion of the patient’s specific situation, goals, risks, and benefits, and other relevant medical discussions.
Statements made by any person(s) in this article are not intended to substitute for this discussion or evaluation or as a guarantee as to outcomes. Examples of treatment outcomes in this article are not intended to convey and warranty, either express or implied, as to outcomes, promises, or benefits from treatment. Whether to accept any treatment by a patient should be assessed by the patient as to the risks and benefits of such procedures and only after consultation with a health care professional.
The use of this article constitutes acknowledgment and acceptance of these limitations and disclaimers. Further dissemination of this information in this site is prohibited without express written approval from newvita.
This article is cited from Vet. Sci. 2022, 9(6), 252; https://doi.org/10.3390/vetsci9060252
The information mentioned in this article is for education and information only. It is not intended to diagnose, treat, cure, or prevent any disease and do not replace medical advice. Advice on treatment or care of an individual patient should be obtained through consultation with a physician or trained health care practitioner who has examined that patient or is familiar with that patient's medical history.
This information and advice published or made available through this website are not intended to replace the services of a physician or a health care professional acting under a physician’s supervision, nor does it constitute a doctor-patient relationship. Each individual’s treatment and/or results may vary based upon the circumstances, the patients’ specific situation, as well as the health care provider’s medical judgment and only after further discussion of the patient’s specific situation, goals, risks, and benefits, and other relevant medical discussions.
Statements made by any person(s) in this article are not intended to substitute for this discussion or evaluation or as a guarantee as to outcomes. Examples of treatment outcomes in this article are not intended to convey and warranty, either express or implied, as to outcomes, promises, or benefits from treatment. Whether to accept any treatment by a patient should be assessed by the patient as to the risks and benefits of such procedures and only after consultation with a health care professional.
The use of this article constitutes acknowledgment and acceptance of these limitations and disclaimers. Further dissemination of this information in this site is prohibited without express written approval from newvita.
*This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.